Two decades of ocean acidification experiments have shown that elevated CO2 can affect many traits in fish early life stages. Only few species, however, show direct CO2-induced survival reductions. This may partly reflect a bias in our current empirical record, which is dominated by species from nearshore tropical-to-temperate environments. There, these organisms already experience highly variable CO2 conditions. In contrast, fishes from more offshore habitats, especially at higher latitudes are adapted to more CO2-stable conditions, which could make them more CO2-sensitive. This group of fishes is still underrepresented in the literature, despite its enormous commercial and ecological importance.
To help address this gap, we conducted new experimental work on northern sand lance Ammodytes dubius, a key forage fish on offshore Northwest Atlantic sand banks with trophic links to more than 70 different predator species of fish, squid, seabirds, and marine mammals. On Stellwagen Bank in the southern Gulf of Maine, sand lance are the ‘backbone’ of the eponymous National Marine Sanctuary.
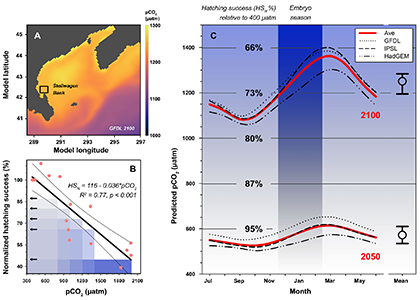
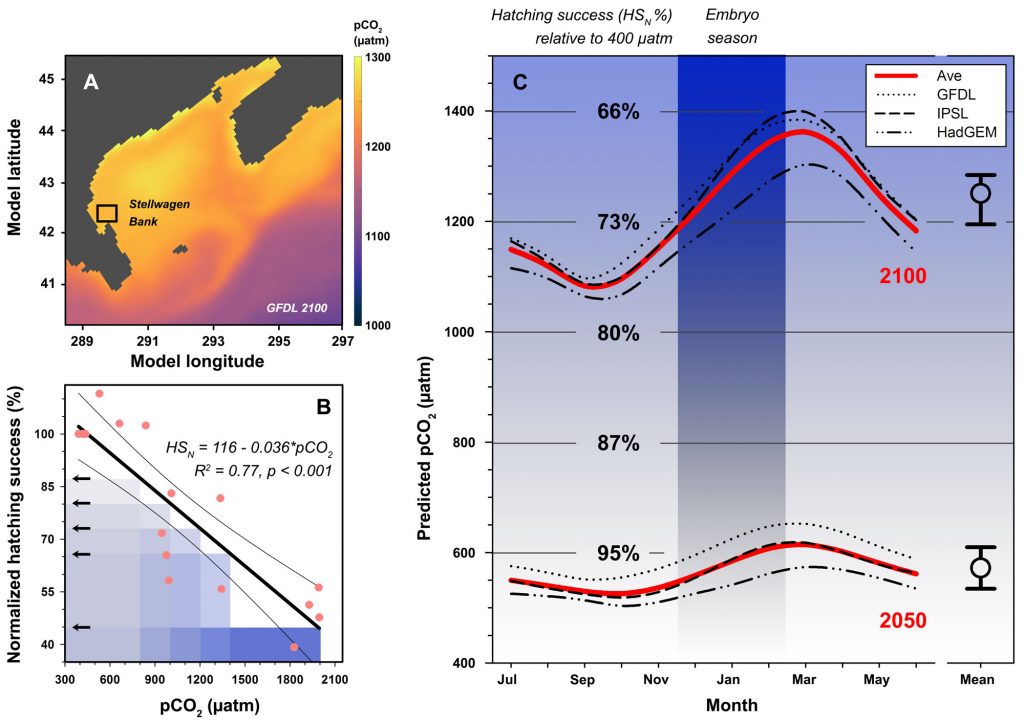
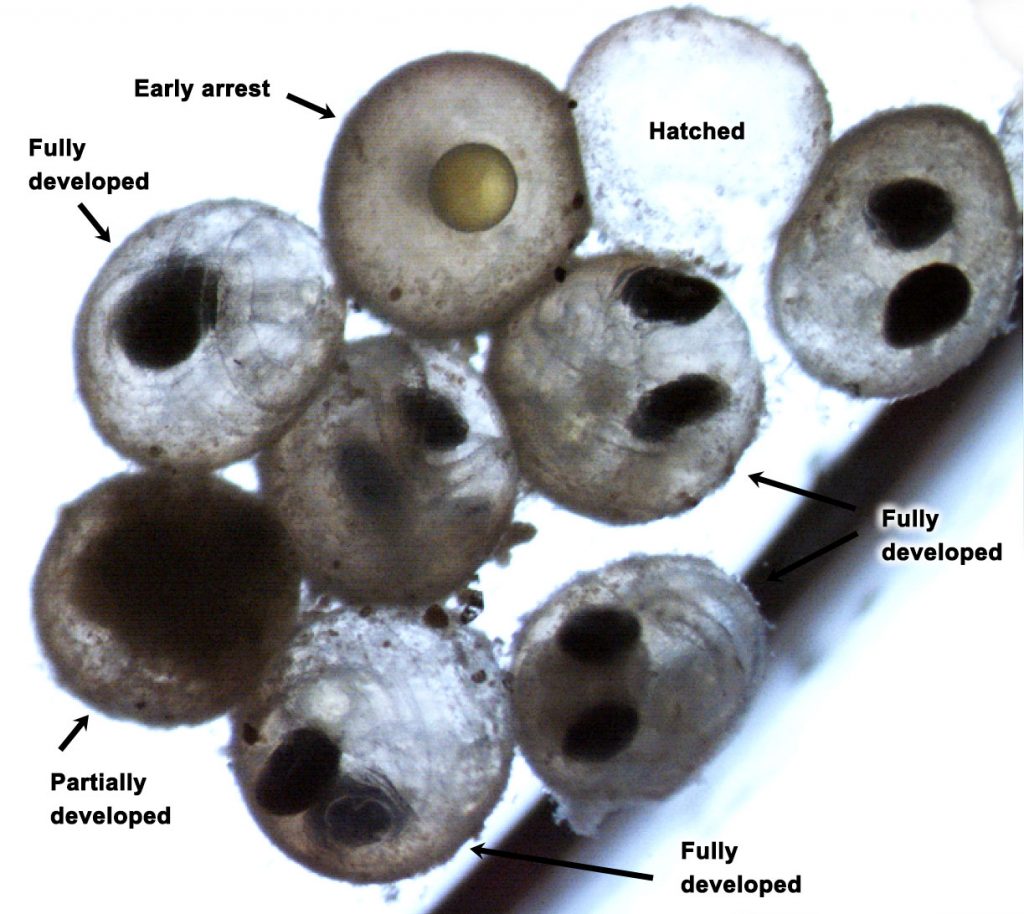
We followed up on the intriguing findings of a pilot study a few years ago. Over two years and two trials, we again produced embryos from wild, Stellwagen Bank spawners and reared them at several pCO2 levels (~400−2000 μatm) in combination with static and dynamic temperatures. Again, we observed consistently large CO2-induced reductions in hatching success (-23% at 1,000 µatm, -61% at ~2,000 µatm), but this time the effects were temperature-independent.
Intriguingly, we again saw that many sand lance embryos at high CO2 treatments did not merely arrest in their development (indicative of acidosis), but appeared to develop fully to hatch but were somehow incapable of doing so. We show several lines of evidence supporting the hypothesis that CO2 directly impairs hatching in this species. Most fish rely on hatching enzymes that help embryos break the chorion (egg shell), but these ubiquitous enzymes may work less efficiently under high CO2, low pH conditions.
For additional context, we also derived long-term, seasonal pCO2 projections specifically for Stellwagen Bank, which together with the experimental data suggested that increasing CO2 levels alone could reduce sand lance hatching success to 71% of contemporary levels by the year 2100.
We believe that the importance of sand lances as forage fishes across most northern hemisphere shelf ecosystem warrants a strategic effort of OA researchers to begin testing other sand lance species or populations to understand the magnitude of the problem and its underlying mechanisms.
Authors:
Hannes Baumann (University of Connecticut)
Lucas Jones (University of Connecticut)
Christopher Murray (University of Washington)
Samantha Siedlecki (University of Connecticut)
Michael Alexander (NOAA Physical Sciences Laboratory)
Emma Cross (Southern Connecticut State University)